A 27-year-old man presents with his second episode of fulminant meningococcal septicaemia. There is no history of immunosuppressive therapy or HIV infection. You suspect an inherited defect of the complement system. Deficiency of which single complement protein most strongly predisposes to disseminated meningococcal infection?
A. C1q
B. C2
C. C3
D. C4
E. C5
Answer
C5



Detailed discussion for MRCP
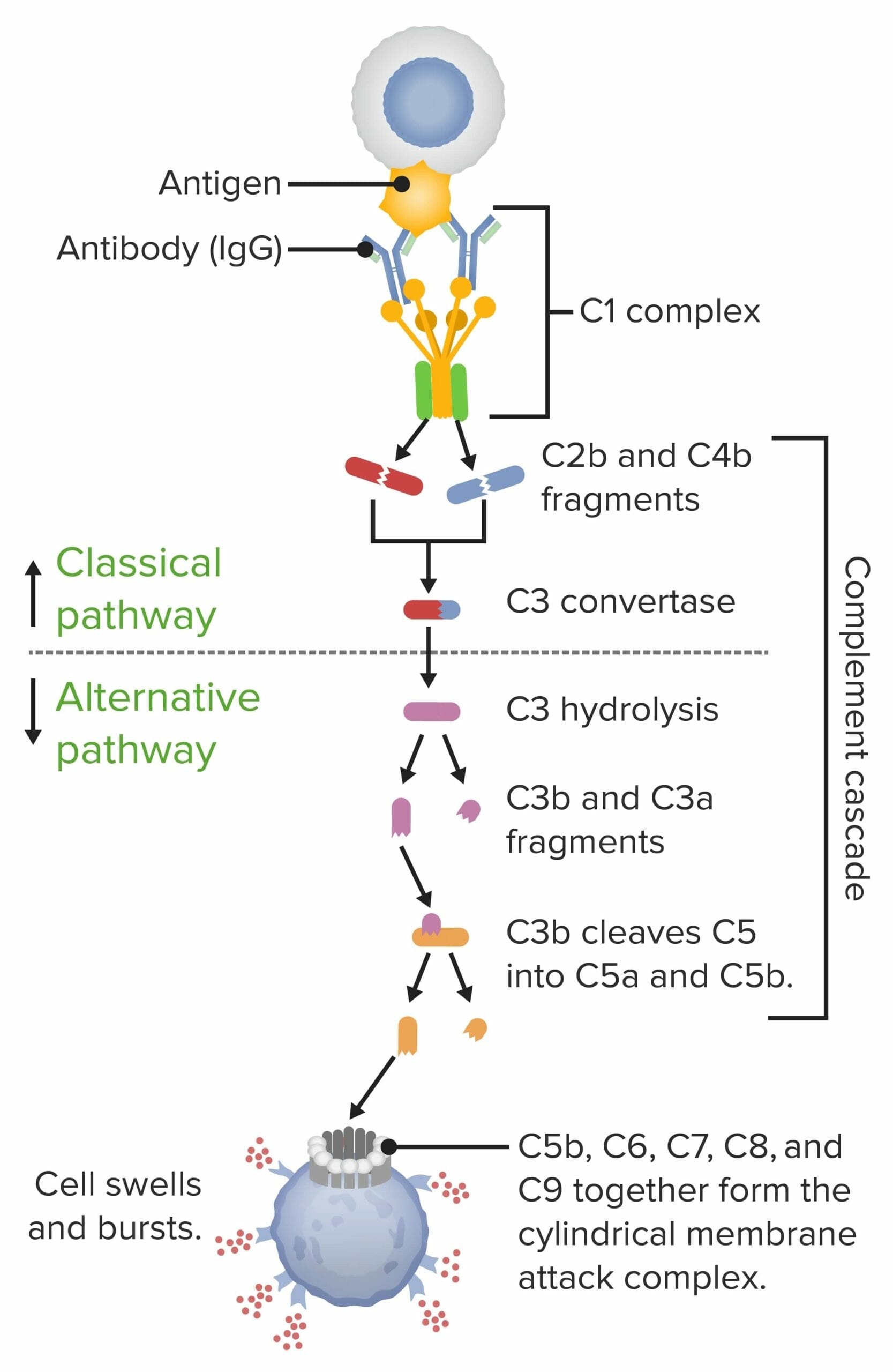
The complement system is a core component of innate immunity and is especially important for defence against gram-negative bacteria, particularly Neisseria meningitidis. Among all complement defects, terminal complement component deficiencies (C5–C9) carry the strongest and most specific association with recurrent and disseminated meningococcal infection.
C5 is pivotal because it is cleaved into C5a and C5b.
• C5a is one of the most potent anaphylatoxins and chemoattractants, recruiting neutrophils and amplifying inflammation.
• C5b initiates formation of the membrane attack complex (MAC) by sequential binding of C6, C7, C8, and C9.
Without C5, the MAC cannot form. Neisseria species are uniquely vulnerable to MAC-mediated killing, so patients with C5 (or C6–C9) deficiency are unable to lyse meningococci efficiently, despite otherwise intact immunity.
Key MRCP-relevant clinical pearls:
- Patients often present later in life (adolescence or adulthood), unlike antibody deficiencies that present in childhood.
- Infections may be recurrent but surprisingly less severe than in asplenia or antibody deficiency, with lower mortality despite frequent episodes.
- Classical signs of immunodeficiency (chronic infections with many organisms) are often absent.
- Vaccination against Neisseria meningitidis is essential, but does not fully eliminate risk.
- Prophylactic antibiotics (e.g. penicillin) may be considered in confirmed cases.
Why the other options are less correct:
- C1q / C4 deficiency → immune complex disease (SLE-like illness), photosensitive rash, glomerulonephritis.
- C2 deficiency (most common complement deficiency) → infections with encapsulated bacteria but weaker meningococcal link.
- C3 deficiency → severe, early-onset, life-threatening pyogenic infections with multiple organisms, not selectively meningococci.
Hence, when MRCP asks about recurrent meningococcal disease, always think terminal complement deficiency, and if one protein is requested, C5.
Complement system — explained simply
Think of the complement system as a built-in security team in the blood that helps the body fight infections. It is part of innate immunity, meaning it is ready to act immediately, even before antibodies are made.
It is called complement because it “complements” (helps) antibodies and white blood cells.
What is the complement system?
-
A group of ~30 proteins present in blood and tissues
-
Mostly made by the liver
-
Normally inactive, but become active in a cascade (one activates the next)
What does the complement system do? (3 core functions)
1. Opsonisation – “tagging the enemy”
-
Complement protein C3b sticks to bacteria
-
This acts like a sticky label
-
Phagocytes (neutrophils, macrophages) see the label and eat the bacteria more easily
👉 Think: “Put a fluorescent jacket on the germ so police can catch it”
2. Inflammation – “calling reinforcements”
-
Small fragments like C3a and C5a are released
-
They:
-
Attract neutrophils
-
Increase blood vessel permeability
-
Cause redness, swelling, heat
-
👉 Think: “Alarm sirens calling immune cells to the site”
3. Direct killing – “punching holes”
-
Late complement proteins (C5–C9) form the Membrane Attack Complex (MAC)
-
MAC punches holes in bacterial cell membranes
-
Especially important for gram-negative bacteria like Neisseria
👉 Think: “Drilling holes in the enemy wall”
How is the complement system activated? (3 pathways)
All pathways have different starts, but same ending.
1. Classical pathway
-
Triggered by antibodies (IgG or IgM) bound to bacteria
-
Uses C1, C2, C4
-
Links adaptive immunity → innate immunity
🧠 Exam tip: Classical pathway = antibodies involved
2. Lectin pathway
-
Triggered when mannose-binding lectin binds sugars on microbes
-
No antibodies needed
-
Uses proteins similar to classical pathway
🧠 Exam tip: Lectin = sugar recognition
3. Alternative pathway
-
Activated directly by microbial surfaces
-
Always slightly “on”
-
Important in early infection
-
Uses Factor B, D, Properdin
🧠 Exam tip: Alternative = antibody-independent, always active
Common final pathway (very important)
All three pathways → activate C3 → then C5–C9 → MAC
👉 That is why C3 is central and C5–C9 are crucial for killing bacteria
High-yield clinical correlations (easy memory)
-
C3 deficiency
→ severe, recurrent bacterial infections from early childhood -
C5–C9 deficiency
→ recurrent meningococcal infections -
C1, C2, C4 deficiency
→ immune complex diseases like SLE -
C1 esterase inhibitor deficiency
→ hereditary angioedema (bradykinin-mediated swelling)
One-line memory summary (gold for exams)
Complement system = proteins that tag bacteria, call immune cells, and punch holes in microbes
Or even shorter:
C3 tags, C5–C9 kills
Complement deficiencies — explained as a story 🌟
Imagine your body is a large fortified city, constantly under threat from invading enemies (bacteria).
The complement system is the city’s automated defence force—guards, alarms, and weapons that activate instantly.
Now, let’s walk through the city and see what happens when different parts of the defence system are missing.
🏰 Scene 1: The broken city gate sensors (C1, C2, C4 deficiency)
At the main gate, there are sensors that detect enemies once antibodies have spotted them.
These sensors are C1, C2, and C4.
One day, these sensors malfunction.
-
Enemies can still be fought
-
But debris (immune complexes) starts piling up inside the city
Soon, the city suffers self-damage:
-
Rashes
-
Joint pain
-
Kidney inflammation
🧠 Medical translation
-
C1, C2, C4 deficiency → immune complex diseases
-
Classically associated with SLE-like illness
-
Infections occur, but are not the main problem
🌪️ Scene 2: The uncontrollable flood (C1 esterase inhibitor deficiency)
In another part of the city is a flood-control system that regulates pressure in water pipes.
This is the C1 esterase inhibitor.
One day, the regulator breaks.
Suddenly, water floods:
-
The face
-
The lips
-
The airway
-
The gut
No itching. No hives. Just terrifying swelling.
🧠 Medical translation
-
C1-INH deficiency → hereditary angioedema
-
Caused by excess bradykinin
-
Does not respond to antihistamines or steroids
-
Airway swelling can be fatal
🧲 Scene 3: The missing “enemy tags” (C3 deficiency)
Now, imagine the city’s guards rely on bright fluorescent tags to recognize enemies.
These tags are C3b.
But the tagging machine breaks completely.
Enemies now:
-
Slip through unnoticed
-
Multiply rapidly
-
Attack again and again from early childhood
🧠 Medical translation
-
C3 deficiency → severe, recurrent bacterial infections
-
Starts in early life
-
Especially encapsulated organisms
-
Can be fatal without treatment
🧠 Exam gold: C3 is central to all pathways
💣 Scene 4: The missing final weapon (C5–C9 deficiency)
Finally, deep inside the city is the ultimate weapon—a laser cannon that drills holes in enemy tanks.
This weapon is the Membrane Attack Complex (C5–C9).
One critical part (C5) is missing.
Now:
-
Most enemies can still be chased away
-
But one enemy keeps returning again and again
That enemy is Neisseria meningitidis.
Strangely:
-
Attacks happen repeatedly
-
But many soldiers survive each attack
🧠 Medical translation
-
C5–C9 deficiency → recurrent meningococcal infections
-
Patients often present in adolescence or adulthood
-
Infections may be recurrent but less fatal
-
Vaccination and prophylactic antibiotics are key
🧠 One-look memory map
| Defect | Story image | Clinical picture |
|---|---|---|
| C1, C2, C4 | Broken sensors | SLE-like disease |
| C1-INH | Flooding pipes | Hereditary angioedema |
| C3 | No enemy tags | Severe childhood infections |
| C5–C9 | No final weapon | Recurrent meningococcal disease |
🏁 Final story takeaway (exam-perfect)
Early components cause autoimmunity, middle component (C3) causes severe infections, and terminal components cause meningococcal disease.
If you remember only one sentence for exams, remember this 👆
Cheat sheet (exam-focused)
- Most specific for meningococcal infection: C5–C9
- Single best answer: C5
- Mechanism: failure of MAC formation
- Organism: Neisseria meningitidis
- Age of presentation: often adolescence/adulthood
- Severity: recurrent but relatively low mortality
- C3 deficiency: severe early childhood infections
- C1/C4 deficiency: SLE-like disease
- C1-INH deficiency: hereditary angioedema (bradykinin-mediated)
Flash cards
1. Q: Which complement components form the MAC?
A: C5–C9
Explanation: MAC causes lysis of gram-negative bacteria.
2. Q: Most common organism in terminal complement deficiency?
A: Neisseria meningitidis
Explanation: Highly susceptible to MAC-mediated killing.
3. Q: Which complement deficiency presents later in life?
A: C5–C9 deficiency
Explanation: Unlike antibody deficiencies.
4. Q: Why is C5 critical?
A: Generates C5a (inflammation) and C5b (MAC initiation).
5. Q: What does C5a do?
A: Potent chemoattractant and anaphylatoxin.
6. Q: What does C5b do?
A: Starts MAC assembly.
7. Q: C3 deficiency causes what pattern of infection?
A: Severe recurrent pyogenic infections early in life.
8. Q: C1q deficiency is linked to which disease?
A: SLE.
9. Q: Most common complement deficiency?
A: C2 deficiency.
10. Q: Encapsulated bacteria risk in C3 deficiency?
A: Very high.
11. Q: Which complement deficiency causes hereditary angioedema?
A: C1 esterase inhibitor deficiency.
12. Q: Mediator responsible for angioedema in C1-INH deficiency?
A: Bradykinin.
13. Q: Does vaccination fully protect terminal complement-deficient patients?
A: No.
14. Q: Mortality of meningococcal infection in terminal complement deficiency?
A: Lower than expected.
15. Q: Why lower mortality?
A: Preserved inflammatory response despite impaired lysis.
16. Q: Screening test for complement deficiency?
A: CH50 (classical pathway).
17. Q: CH50 low, AH50 normal suggests?
A: Classical pathway defect.
18. Q: CH50 and AH50 both low suggests?
A: Terminal complement deficiency.
19. Q: Organisms classically killed by MAC?
A: Gram-negative bacteria.
20. Q: One-line MRCP association for recurrent meningococcal disease?
A: Think C5–C9 deficiency.
MCQs to test yourself
1. Recurrent meningococcal septicaemia is most strongly associated with deficiency of:
A. C1q
B. C2
C. C3
D. C4
E. C5
Answer: E – loss of MAC formation.
2. The membrane attack complex begins with:
A. C3b
B. C4b
C. C5b
D. C6
E. C9
Answer: C – C5b initiates MAC.
3. Which complement deficiency presents earliest in life?
A. C1q
B. C2
C. C3
D. C5
E. C8
Answer: C – severe early infections.
4. SLE-like disease is associated with deficiency of:
A. C5
B. C1q
C. C8
D. Properdin
E. Factor B
Answer: B – immune complex disease.
5. Most potent anaphylatoxin:
A. C3b
B. C4a
C. C5a
D. C5b
E. C9
Answer: C – strongest chemoattractant.
6. Organism most dependent on MAC for clearance:
A. Streptococcus pneumoniae
B. Staphylococcus aureus
C. Haemophilus influenzae
D. Neisseria meningitidis
E. Listeria monocytogenes
Answer: D.
7. Normal immunoglobulins but recurrent meningococcal disease suggests:
A. Asplenia
B. HIV
C. Terminal complement deficiency
D. CVID
E. SCID
Answer: C.
8. CH50 low, AH50 low indicates:
A. Classical pathway defect
B. Alternative pathway defect
C. Terminal pathway defect
D. Lectin pathway defect
E. Antibody deficiency
Answer: C.
9. C1-INH deficiency leads to:
A. Histamine-mediated angioedema
B. Bradykinin-mediated angioedema
C. Urticaria
D. Mast cell degranulation
E. IgE excess
Answer: B.
10. Best long-term preventive strategy in C5 deficiency:
A. Steroids
B. IVIG
C. Vaccination + possible antibiotic prophylaxis
D. Splenectomy
E. Interferon
Answer: C.
Now let us do some MCQs to test our knowledge of complement system.
All questions are in the format- which of the following statements is FALSE?
1. Regarding the complement system:
A. It is part of innate immunity
B. Most complement proteins are synthesised by the liver
C. Complement proteins circulate in inactive forms
D. Complement activation occurs via cascade amplification
E. Complement acts only after T-cell activation
Answer: E
Explanation: Complement is part of innate immunity and acts immediately.
2. Regarding opsonisation:
A. C3b is the major opsonin
B. Opsonisation enhances phagocytosis
C. Neutrophils have receptors for C3b
D. Opsonisation reduces bacterial killing
E. It improves recognition of pathogens
Answer: D
Explanation: Opsonisation enhances, not reduces, bacterial killing.
3. Regarding complement-mediated inflammation:
A. C3a and C5a are anaphylatoxins
B. C5a is a strong neutrophil chemoattractant
C. Complement increases vascular permeability
D. Complement reduces leukocyte recruitment
E. Complement promotes inflammatory response
Answer: D
Explanation: Complement increases leukocyte recruitment.
4. Regarding the membrane attack complex (MAC):
A. It consists of C5–C9
B. It causes pore formation in cell membranes
C. It is particularly important against gram-negative bacteria
D. It is essential for opsonisation
E. It causes direct bacterial lysis
Answer: D
Explanation: Opsonisation is mediated mainly by C3b, not MAC.
5. Regarding the classical complement pathway:
A. It requires antibodies for activation
B. IgG and IgM can activate it
C. It involves C1, C2, and C4
D. It is antibody-independent
E. It links adaptive and innate immunity
Answer: D
Explanation: Classical pathway is antibody-dependent.
6. Regarding the lectin pathway:
A. It is activated by microbial sugars
B. Mannose-binding lectin is involved
C. Antibodies are required
D. It converges at C3
E. It resembles the classical pathway
Answer: C
Explanation: Lectin pathway does not require antibodies.
7. Regarding the alternative pathway:
A. It is continuously active at low levels
B. It is antibody-independent
C. It uses factor B and factor D
D. It is the earliest defence mechanism
E. It requires C1 activation
Answer: E
Explanation: C1 is not involved in the alternative pathway.
8. Regarding C3:
A. It is central to all complement pathways
B. Its deficiency causes severe infections
C. It plays a role in opsonisation
D. Its deficiency presents late in adulthood
E. It is essential for MAC formation
Answer: D
Explanation: C3 deficiency presents early in life.
9. Regarding C3 deficiency:
A. It presents in early childhood
B. It causes recurrent pyogenic infections
C. It predisposes to encapsulated organisms
D. It is usually mild and self-limiting
E. It can be life-threatening
Answer: D
Explanation: C3 deficiency is severe, not mild.
10. Regarding terminal complement deficiency (C5–C9):
A. It predisposes to meningococcal infection
B. MAC formation is impaired
C. Patients often present in adolescence or adulthood
D. Mortality is always extremely high
E. Recurrent infections are characteristic
Answer: D
Explanation: Mortality is often lower than expected despite recurrence.
11. Regarding Neisseria meningitidis infection:
A. It is a gram-negative organism
B. It is highly susceptible to MAC-mediated killing
C. It commonly infects patients with C5–C9 deficiency
D. It is mainly cleared by antibodies alone
E. Recurrent infection suggests complement deficiency
Answer: D
Explanation: MAC-mediated killing is crucial.
12. Regarding C1, C2, and C4 deficiencies:
A. They affect the classical pathway
B. They predispose to immune complex disease
C. They are associated with SLE-like illness
D. They cause severe early-life pyogenic infections
E. Autoimmunity is a major feature
Answer: D
Explanation: Severe early infections are typical of C3 deficiency.
13. Regarding immune complex diseases:
A. They occur due to poor clearance of immune complexes
B. They are associated with early complement deficiencies
C. SLE is an example
D. They are typical of terminal complement deficiency
E. They involve deposition in tissues
Answer: D
Explanation: Terminal complement deficiency is linked to meningococcal infection, not immune complex disease.
14. Regarding C1 esterase inhibitor deficiency:
A. It causes hereditary angioedema
B. It is mediated by bradykinin
C. It causes urticaria and itching
D. It can cause life-threatening airway swelling
E. Antihistamines are ineffective
Answer: C
Explanation: There is no urticaria or itching.
15. Regarding hereditary angioedema:
A. It is not IgE-mediated
B. It responds well to steroids
C. Bradykinin levels are increased
D. Facial and laryngeal swelling can occur
E. Complement regulation is defective
Answer: B
Explanation: Steroids are ineffective.
16. Regarding complement screening tests:
A. CH50 assesses the classical pathway
B. Low CH50 suggests complement deficiency
C. Normal CH50 excludes all complement defects
D. Terminal pathway defects lower CH50
E. Screening helps identify inherited deficiencies
Answer: C
Explanation: Some defects may still have normal screening tests.
17. Regarding MAC:
A. It is formed after C5 cleavage
B. C9 forms the pore
C. It acts mainly on gram-positive bacteria
D. It causes osmotic lysis
E. It is ineffective in C5 deficiency
Answer: C
Explanation: MAC mainly targets gram-negative bacteria.
18. Regarding complement and age of presentation:
A. Early complement deficiencies present earlier
B. C3 deficiency presents in childhood
C. Terminal complement deficiency may present later
D. Age of onset helps localise the defect
E. All complement deficiencies present in infancy
Answer: E
Explanation: Terminal deficiencies often present later.
19. Regarding vaccination in terminal complement deficiency:
A. It is strongly recommended
B. It reduces infection risk
C. It completely eliminates meningococcal risk
D. It is part of long-term management
E. Antibiotic prophylaxis may be considered
Answer: C
Explanation: Vaccination does not completely eliminate risk.
20. High-yield exam associations:
A. C3 deficiency → severe recurrent infections
B. C1/C4 deficiency → SLE-like disease
C. C5–C9 deficiency → meningococcal infection
D. C1-INH deficiency → histamine-mediated angioedema
E. Complement deficiencies affect innate immunity
Answer: D
Explanation: Hereditary angioedema is bradykinin-mediated, not histamine-mediated.
Summary for quick exam revision
Terminal complement component deficiencies, especially C5–C9, are classically associated with recurrent disseminated meningococcal infection. C5 is essential for both inflammatory amplification (via C5a) and initiation of the membrane attack complex (via C5b). Loss of MAC formation leaves patients unable to lyse Neisseria meningitidis, despite otherwise preserved immunity. These patients often present in adolescence or adulthood and may have recurrent episodes with relatively low mortality. In contrast, C3 deficiency causes severe early-onset pyogenic infections, while C1q, C2, and C4 deficiencies are linked to immune-complex diseases such as SLE. C1-INH deficiency causes hereditary angioedema via bradykinin excess. For MRCP, recurrent meningococcal disease should immediately trigger the association: think terminal complement deficiency.